January 23, 2018 PR-M01-18-NI-068
TUCSON, Ariz., Jan. 16, 2018 /PRNewswire/ — Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced the launch of the CE-marked VENTANA MMR IHC Panel, which provides clinicians with a comprehensive group of immunohistochemistry (IHC) tests for patients diagnosed with colorectal cancer. The tests detect certain proteins associated with a DNA repair mechanism called mismatch repair (MMR), and aid in differentiating between sporadic colorectal cancer and probable Lynch syndrome, a hereditary form of colorectal cancer. About 3 percent of colorectal cancers are associated with Lynch syndrome.2
Lynch syndrome results in a 50 to 80 percent lifetime risk of developing colorectal cancer,3,4 making it important to identify the syndrome in colorectal cancer patients and at-risk family members. Identification of the syndrome may result in early detection and possible cancer prevention among those with the inherited mutation.5
The global burden of colorectal cancer is expected to increase by 60 percent to more than 2.2 million new cases and 1.1 million deaths by 2030.6 Increasingly, guidelines, including those from the National Institute for Health and Care Excellence (NICE), are recommending universal screening of all newly diagnosed colorectal cancers for Lynch syndrome.
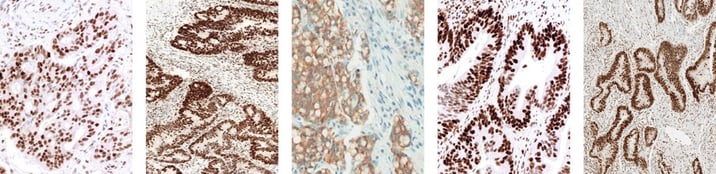 Colorectal cancer staining positive using the VENTANA MMR IHC Panel
Colorectal cancer staining positive using the VENTANA MMR IHC Panel
"This testing impacts not just the patient, but family members who may benefit from further genetic testing and advanced monitoring to detect colorectal cancer at its earlier stages, when it is more treatable," said Ann Costello, Head of Roche Tissue Diagnostics. "The VENTANA MMR IHC Panel provides clinicians with an additional tool to perform universal tumor screening for probable Lynch syndrome as recommended by medical guidelines."
The VENTANA MMR IHC Panel includes four assays that target MMR proteins MLH1, PMS2, MSH2 and MSH6, as well as the VENTANA BRAF V600E (VE1) assay. It is available for use on the BenchMark GX, XT and ULTRA instruments.
About the VENTANA MMR IHC Panel
The CE-marked VENTANA MMR IHC Panel consists of VENTANA anti-MLH1 (M1), VENTANA anti-PMS2 (A16-4), VENTANA anti-MSH2 (G219-1129), VENTANA anti-MSH6 (SP93) antibodies, for patients diagnosed with colorectal cancers for the detection of mismatch repair protein deficiency as an aid in the identification of probable Lynch syndrome, and VENTANA BRAF V600E (VE1) antibody as an aid to differentiate between sporadic colorectal cancer and probable Lynch syndrome.
The inclusion of the VENTANA BRAF V600E (VE1) antibody helps refine the number of patients who may receive additional testing for Lynch syndrome by aiding in the differentiation between sporadic colorectal cancer and probable Lynch syndrome in the absence of MLH1 protein expression.
The ready-to-use VENTANA MMR IHC Panel is optimized for use with the OptiView DAB IHC Detection Kit, OptiView Amplification Kit and ancillaries on the fully-automated BenchMark GX, XT and ULTRA instruments.
References
1This product is intended for in vitro diagnostic (IVD) use
2Sehgal R, Sheahan K, O'Connell PR et al. Lynch syndrome: an updated review. Genes (Basel), 2014;5:497–507
3Buza N, Ziai J, Hui P. Mismatch repair deficiency testing in clinical practice, Expert Review of Molecular Diagnostics, 2016;16:591-604
4Connell LC, Mota JM, Braghiroli MI et al. The Rising Incidence of Younger Patients With Colorectal Cancer: Questions About Screening, Biology, and Treatment, Current Treatment Options in Oncology. 2017;18:23
5Provenzale D, Gupta S, Ahnen DJ, et al. Genetic/Familial High-Risk Assessment: Colorectal Version 1.2016, NCCN Clinical Practice Guidelines in Oncology. J National Comprehensive Cancer Network. 2016;14:1010–1030
6Arnold M, Sierra MS, Laversanne M, et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683-691.
SOURCE Roche
Contact
Roche Tissue Diagnostics Media Relations
Gabrielle Fimbres
Senior Manager, External Communications
Tel: +1 520 222 4573
Email: gabrielle.fimbres@roche.com
ventana.com

Nice Insight, established in 2010, is the research division of That’s Nice, A Science Agency, providing data and analysis from proprietary annual surveys, custom primary qualitative and quantitative research as well as extensive secondary research. Current annual surveys include The Nice Insight Contract Development & Manufacturing (CDMO/CMO), Survey The Nice Insight Contract Research - Preclinical and Clinical (CRO) Survey, The Nice Insight Pharmaceutical Equipment Survey, and The Nice Insight Pharmaceutical Excipients Survey.
